For decades, the medical approach to autoimmune diseases has been one of management rather than resolution. Because the immune system mistakenly identifies the body’s own tissues as threats, treatments have focused on suppressing the immune response. While this helps control symptoms, it leaves the underlying cause intact.
However, a revolutionary shift is underway. By repurposing a breakthrough cancer therapy, scientists are now attempting to eliminate the “rogue” cells responsible for these disorders, moving closer to what could be a definitive cure.
The Cancer Connection: A Shared Biological Flaw
The breakthrough lies in the realization that autoimmune diseases and certain cancers share a fundamental biological mechanism: uncontrolled cell growth.
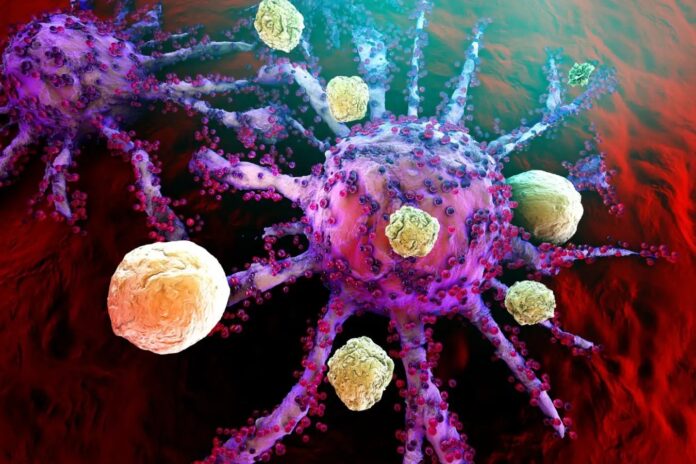
In cancer, mutant cells divide without stopping. In autoimmune conditions—such as Type 1 diabetes, multiple sclerosis, or lupus—rogue immune cells fail their internal “screening” process. Recent studies suggest these rogue cells possess genetic mutations that prevent them from self-destructing when they mistakenly target healthy organs. Essentially, these cells behave like a slow-motion version of cancer, persistently attacking the body’s own insulin-producing cells or nerve sheaths.
Adapting the “Living Drug”: CAR T-Cell Therapy
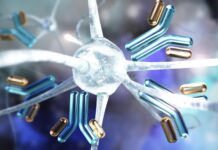
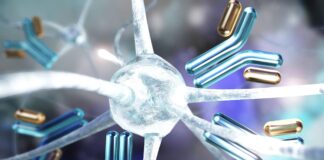
The technology at the heart of this movement is CAR T-cell therapy. Originally designed to fight blood cancers, this process involves:
1. Extracting T-cells from a patient’s blood.
2. Genetically engineering them to recognize and attack specific targets.
3. Reintroducing them into the patient to hunt down the diseased cells.
While highly effective in oncology, CAR T-cells have historically been plagued by two major hurdles: they primarily work on blood cancers (not solid tumors) and can cause severe side effects, such as brain inflammation.
The “Miraculous” Discovery in Autoimmune Trials
Recent clinical applications in autoimmune patients have yielded results that surprised the medical community. Researchers at the University Hospital of Erlangen in Germany, led by Fabian Müller, applied CAR T-cell therapy to patients with lupus, and the outcomes were unexpected:
- Self-Limiting Action: Unlike in cancer patients, where CAR T-cells persist for years, in autoimmune patients, the cells appear to do their job and then disappear after a few months.
- Safer Profile: The life-threatening side effects seen in cancer treatments have not been observed in these autoimmune trials.
- Immune Recovery: Because the CAR T-cells eventually clear out of the system, the patient’s immune system is able to reset and recover.
“We just were lucky, honestly,” says Müller, noting that the unexpected disappearance of the cells changes the risk-benefit analysis, making the treatment viable for a much broader range of patients.
Challenges on the Horizon: Cost and Complexity
Despite the optimism, several hurdles remain before this becomes a standard of care for the 1 in 10 people affected by autoimmune disorders:
- Irreversible Damage: While the therapy can stop the immune attack, it cannot always repair the damage already done to organs (such as the pancreas in diabetes or nerves in MS).
- Extreme Costs: The current “bespoke” model—where cells are custom-made for every single patient—is prohibitively expensive.
- Targeting Precision: It is difficult to kill only the “rogue” cells without destroying the healthy antibody-producing cells needed for general immunity.
The Future: “Off-the-Shelf” and “In Vivo” Solutions
To make this treatment accessible, the industry is moving toward two innovative models:
– “Off-the-shelf” CAR T-cells: Using donor cells that can be mass-produced for many patients. Interestingly, while these haven’t been as effective in cancer, they may be perfectly suited for the shorter-term needs of autoimmune patients.
– “In vivo” CAR T-cells: A futuristic approach where cells are engineered inside the patient’s body, potentially eliminating the need for expensive lab processes entirely.
Conclusion
By shifting the goal from suppressing symptoms to eliminating rogue cells, CAR T-cell therapy offers a glimpse of a future where autoimmune diseases are no longer lifelong burdens but treatable conditions. While high costs and biological complexities remain, the clinical success seen so far marks a historic turning point in immunology.